By: Dr. Coni Horndli PhD.
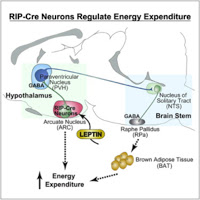
This recent study in Cell from Brad Lowell’s group reveals novel circuits in the arcuate nucleus of the hypothalamus that selectively drive energy expenditure. The authors integrate genetic and electrophysiological approaches with studies of metabolism and neuronal circuit mapping to reveal a defined population of GABAergic neurons that regulate brown fat and energy expenditure. It remarkable to see such potent neural control over these aspects of physiology.
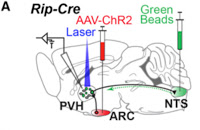
The authors use an elegant double-labeling method to reveal functional neuronal connectivity between RIP-Cre ARC – PVH – NTS neurons. First, a Cre-dependent virus, which expresses a channelrhodopsin-mCherry fusion protein (AAV-Flex-ChR2(H134R)-mCherry) was stereotaxically injected in the ARC of RIP-Cre mice. Second, retrograde green fluorescent beads were injected into the NTS of the same mice. Electrophysiological recordings specifically showed light-evoked IPSCs in bead+ PVH neurons.
In this study, a synthetic receptor was expressed in RIP-Cre mice and activated by the pharmacological compound CNO. Neuronal activity in response to CNO and later leptin were recorded in cultured brain slices. It would be interesting to see whether the same neuronal responses can be elicited using an in vivo approach.
In the ARC, only 30% of RIP-Cre neurons showed an excitatory response to leptin, while 35% of ARC RIP-Cre neurons were inhibited. If and how ARC neurons exhibiting these differential responses integrate into one circuit and or what the role of leptin-inhibited ARC RIP-Cre neurons is in regulating energy expenditure is not clear at this point. Also, I am wondering how, where and when AgRP, which reside in the ARC, project to the PVH and are also GABAergic but inhibit energy expenditure and food intake interplay in the RIP-Cre ARC – PVH – NTS circuitry.
REFERENCE AND ABSTRACT:
GABAergic RIP-Cre Neurons in the Arcuate Nucleus Selectively Regulate Energy Expenditure
Dong Kong, Qingchun Tong, Chianping Ye, Shuichi Koda, Patrick M. Fuller, Michael J. Krashes, Linh Vong,Russell S. Ray, David P. Olson, and Bradford B. Lowell
SUMMARY
Neural regulation of energy expenditure is incompletely understood. By genetically disrupting GABAergic transmission in a cell-specific fashion, and by combining this with selective pharmacogenetic activation and optogenetic mapping techniques, we have uncovered an arcuate-based circuit that selectively drives energy expenditure. Specifically, mice lacking synaptic GABA release from RIP-Cre neurons have reduced energy expenditure, become obese and are extremely sensitive to highfat diet-induced obesity, the latter due to defective diet induced thermogenesis. Leptin’s ability to stimulate thermogenesis, but not to reduce feeding, is markedly attenuated. Acute, selective activation of arcuate GABAergic RIP-Cre neurons, which monosynaptically innervate PVH neurons projecting to the NTS, rapidly stimulates brown fat and increases energy expenditure but does not affect feeding. Importantly, this response is dependent upon GABA release from RIP-Cre neurons. Thus, GABAergic RIP-Cre neurons in the arcuate selectively drive energy expenditure, contribute to leptin’s stimulatory effect on thermogenesis, and protect against diet-induced obesity.